Race is now on to protect those at risk
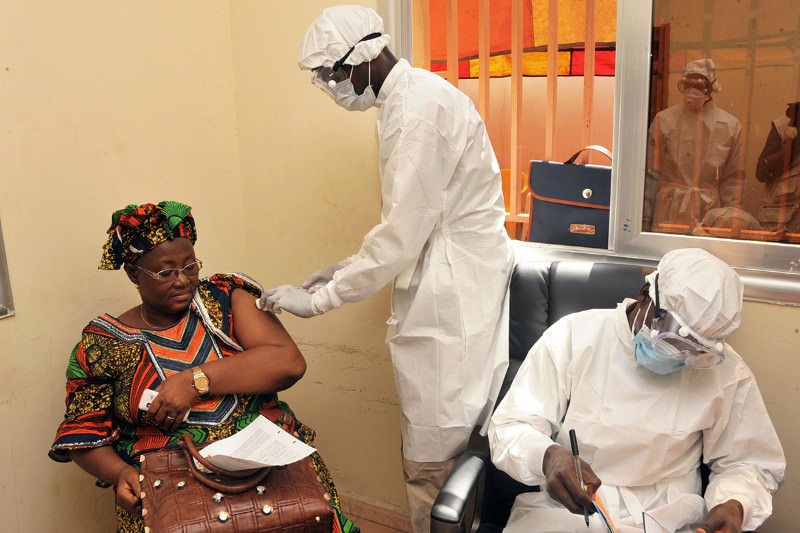
A vaccine can now protect against Ebola (Image: Cellou BinaniI/AFP/Getty Images)A vaccine for Ebola produced in just one year instead of the usual decade provides 100-per-cent protection against the disease. Preparations are already under way to make it available to healthcare workers and families wherever the virus remains at large.
“This is a very good day,” says Seth Berkley, chief executive of the GAVI Alliance, the global organisation that has earmarked $390 million to extend availability of the VSV-ZEBOV vaccine beyond Guinea, the country where it has been successfully tested on more than 7500 people.
The epidemic is now largely under control, but there have been 28,000 cases leading to 11,000 deaths in Guinea, Sierra Leone and Liberia. But cases are still coming to light, and the virus is still at large, lurking in the body fluids of survivors for as long as 6 months.
“All affected countries should immediately start and multiply ring vaccinations to break chains of transmission and vaccinate all frontline workers to protect them,” says Bertrand Draguez, medical director of relief organisation Médecins Sans Frontières.
The rapid availability of a vaccine would be a huge boost for citizens of those three countries and for all the health workers still operating in them to deal with new cases.
Emergency authorisation
Berkley says that the GAVI Alliance has already earmarked “considerable” funds that could make this roll out possible, and that discussions are under way with the governments of affected countries, the funders of the clinical trials, the manufacturer of the vaccine and the World Health Organization to decide how to move forward as fast as possible.
Although the trial isn’t yet over, the WHO could theoretically issue an Emergency Use Authorisation before it ends. This would enable the vaccine to be legally deployed where needed.
“When there’s a WHO recommendation, we will be willing to purchase and stockpile the vaccine until we get regulatory authorisation from individual governments,” Berkley says.
The trial is still in progress, but evidence that the vaccine works arose by studying how well the vaccine protected health workers and family members associated with new cases of Ebola. They wanted to see how well this strategy of “ring vaccination” – immunising those people in closest contact with any new cases – protected those exposed.
Rather than vaccinating only half the participants, and risking the lives of others by giving them a dummy vaccine, the researchers gave half the participants the vaccine immediately after they had contact with a newly diagnosed case, while the other half received it three weeks later.
After three months, the result was resounding. No instant recipients were infected, but 16 of those receiving the vaccine three weeks later were. “This is an extremely promising development,” said Margaret Chan, director-general of the WHO. The results at three months were so impressive that all subsequent participants have since been receiving the vaccine immediately.
Hard to store
Developed initially by the Public Health Agency of Canada, the vaccine is a live but harmless virus called vesicular stomatitis virus, which normally infects animals, engineered to contain a key fragment from the Ebola virus. The immune systems of recipients make antibodies that prime for defence against the real virus.
The race is now on to provide it wherever possible, pending negotiations between all the parties involved. “We can say that Guinea is now taken care of, but what happens in Sierra Leone and Liberia, and other countries where there are outbreaks such as Mali and Uganda,” says Berkley. “We need some type of procedure to say it’s OK to use it in these other settings.”
Berkley said that the vaccine is not perfect. It must be stored at -80 °C, which is not easy to guarantee in tropical countries. It also may not protect against all strains of Ebola. Nor is it yet established how long immunity lasts.
But other trials of vaccines against ebola are under way, says Margaret Harris of the WHO. In Liberia, the VSV-ZEBOV vaccine is being trialled head-to-head against a rival vaccine produced by Glaxo-Smith Kline, and a vaccine developed by the US Centers for Disease Control and Prevention in Atlanta, Georgia, is being tested in Sierra Leone.
Harris said that cases are still coming to light, but they are waning in number. “We’ve had a very encouraging week, and for the first time we’ve seen a column of zeros where there are usually new cases.”
Journal reference: The Lancet, DOI: 10.1016/S0140-6736(15)61117-5
